Prostate Cancer and You:
A Guide for Patients and Caregivers
This comprehensive guide was designed to work in tandem with you and your physician to answer your questions and provide guidance on how to live well during your prostate cancer treatment.
A Comprehensive Resource for those Undergoing Treatment for Prostate Cancer
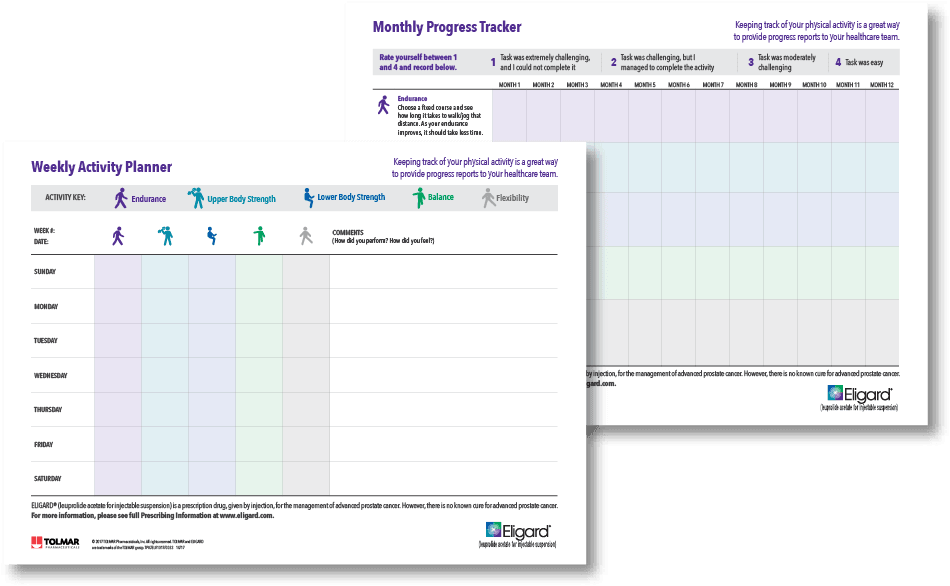
Learn about prostate cancer, diagnosis, management and treatment options. Including, easy-to-use tools for tracking physical activity and recording questions as well as symptoms and side effects.
Important Safety Information
ELIGARD® is indicated for the palliative treatment of advanced prostate cancer.
ELIGARD® is contraindicated in patients with hypersensitivity to GnRH, GnRH agonist analogs, or any of the components of ELIGARD. Anaphylactic reactions to synthetic GnRH or GnRH agonist analogs have been reported in the literature. Transient increase in serum levels of testosterone during treatment may result in worsening of symptoms or onset of new signs and symptoms during the first few weeks of treatment, including bone pain, neuropathy, hematuria, bladder outlet obstruction, ureteral obstruction, or spinal cord compression.
Hyperglycemia and an increased risk of developing diabetes have been reported in men receiving GnRH analogs. Monitor blood glucose level and manage according to current clinical practice. Increased risk of myocardial infarction, sudden cardiac death and stroke has also been reported with use of GnRH analogs in men. Monitor for cardiovascular disease and manage according to current clinical practice. Androgen deprivation therapy may prolong the QT/QTc interval. Consider risks and benefits. May cause fetal harm. Convulsions have been observed in patients taking leuprolide acetate with or without a history of predisposing factors. Manage convulsions according to current clinical practice.
ELIGARD may impair fertility in males of reproductive potential.
The most common injection site adverse events are transient burning and stinging, pain, bruising, and erythema. The most common systemic adverse events include mild to severe hot flashes/sweats, malaise and fatigue, weakness, myalgia, dizziness, clamminess, testicular atrophy, and gynecomastia. As with other GnRH agonists, other adverse reactions, including decreased bone density and rare cases of pituitary apoplexy have been reported. Click for full prescribing and safety information .